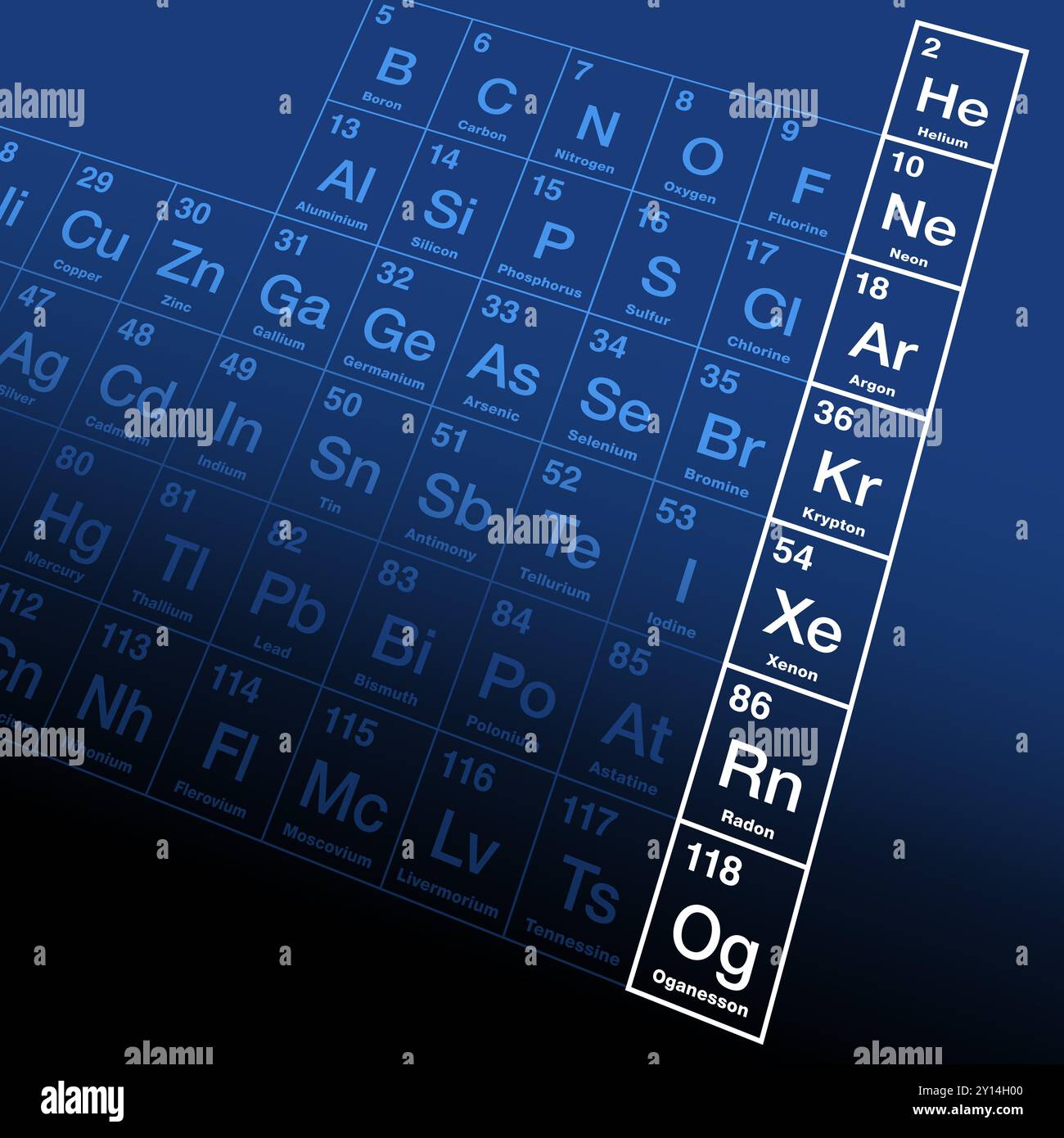
The Noble Gas Periodic Table is a enamour subject that delves into the unequalled properties and behaviors of the elements known as noble gases. These gases, which include helium, neon, argon, krypton, xenon, and radon, are located in Group 18 of the periodical table. Their constancy and inertness create them crucial in various scientific and industrial applications. This post will explore the characteristics, uses, and import of noble gases, providing a comprehensive translate of their role in the periodic table.
The Characteristics of Noble Gases
The noble gases are characterize by their total outer electron shells, which make them exceedingly stable and non reactive. This constancy is a key divisor in their chemical behavior and applications. Let's delve into the specific properties of each noble gas:
- Helium (He): The lightest and most abundant noble gas, helium is known for its use in balloons and airships due to its low density. It is also used in cryogenics and as a shielding gas in weld.
- Neon (Ne): Neon is famous for its use in neon signs, where it emits a distinctive red orange glow when electrically excite. It is also used in high voltage indicators and advertising signs.
- Argon (Ar): Argon is the third most abundant gas in the Earth's atmosphere. It is usually used in welding and metalworking to create an inert atmosphere, preventing oxidation.
- Krypton (Kr): Krypton is used in energy efficient windows and fluorescent lamps. Its phantasmal lines are used as a standard for length measurement.
- Xenon (Xe): Xenon has several applications, including use in car headlights, aesculapian figure, and as an anesthetic. Its power to form compounds, unlike other noble gases, makes it unique.
- Radon (Rn): Radon is a radioactive gas that is a byproduct of the decay of radium. It is known for its health risks, particularly in indoor environments where it can accumulate and pose a risk of lung crab.
The Historical Discovery of Noble Gases
The discovery of noble gases is a story of scientific peculiarity and perseverance. The first noble gas to be discover was helium, which was initially detected in the Sun's spectrum during a solar eclipse in 1868. It was later isolated on Earth in 1895. The other noble gases were discovered in the late 19th and early 20th centuries through the act of scientists like William Ramsay and Lord Rayleigh, who identify argon, neon, krypton, and xenon. Radon was discovered in 1900 by Friedrich Ernst Dorn.
The Role of Noble Gases in the Periodic Table
The noble gases occupy a unparalleled position in the Noble Gas Periodic Table. They are put in Group 18, the far right column of the periodical table. This placement reflects their full valency electron shells, which give them their characteristic inertness. The periodic table's structure helps in understanding the trends and properties of these elements. for representative, as you displace down the group, the nuclear radius increases, and the boil and melting points also increase.
Here is a table summarizing the key properties of the noble gases:
| Element | Symbol | Atomic Number | Atomic Mass (u) | Boiling Point (C) | Melting Point (C) |
|---|---|---|---|---|---|
| Helium | He | 2 | 4. 0026 | 268. 93 | 272. 2 |
| Neon | Ne | 10 | 20. 180 | 246. 08 | 248. 59 |
| Argon | Ar | 18 | 39. 948 | 185. 85 | 189. 3 |
| Krypton | Kr | 36 | 83. 798 | 153. 22 | 157. 36 |
| Xenon | Xe | 54 | 131. 293 | 108. 1 | 111. 8 |
| Radon | Rn | 86 | 222. 018 | 61. 7 | 71. 0 |
Note: The properties list above are guess values and can vary somewhat count on the source.
Applications of Noble Gases
The singular properties of noble gases make them invaluable in assorted applications. Here are some of the key uses:
- Industrial Applications: Noble gases are used in welding and metalworking to make an inert atmosphere, prevent oxidation and contamination. Argon is especially democratic for this purpose.
- Medical Applications: Xenon is used as an anesthetic due to its ability to induce anesthesia without the side effects of traditional anesthetics. It is also used in aesculapian project.
- Lighting and Display: Neon and argon are used in lighting applications, such as neon signs and fluorescent lamps. Xenon is used in high intensity discharge lamps and car headlights.
- Scientific Research: Noble gases are used in various scientific experiments and enquiry due to their inertness and constancy. for instance, helium is used in cryogenics to achieve extremely low temperatures.
- Environmental Monitoring: Radon detection is essential for environmental monitor, as high levels of radon in indoor environments can pose important health risks.
Safety and Handling of Noble Gases
While noble gases are generally inert and non toxic, handling them requires caution. Here are some safety guidelines:
- Ventilation: Ensure proper airing when plow noble gases to prevent the accruement of gases, which can displace oxygen and result to asphyxiation.
- Personal Protective Equipment (PPE): Use seize PPE, include gloves and safety glasses, to protect against potential hazards.
- Storage: Store noble gases in good ventilated areas away from heat sources and incompatible substances.
- Disposal: Dispose of noble gases agree to local regulations and guidelines to minimize environmental impact.
Note: Always refer to the Material Safety Data Sheet (MSDS) for specific handling and safety instructions for each noble gas.
Noble gases are a fascinating group of elements with alone properties and a wide range of applications. Their constancy and inertness make them essential in diverse scientific and industrial fields. Understanding the characteristics and uses of noble gases provides a deeper taste for their role in the Noble Gas Periodic Table and their significance in mod engineering and enquiry.
From their historical discovery to their modern applications, noble gases continue to play a important role in our read of chemistry and the natural world. Their unique properties make them essential in fields ranging from medicine to environmental supervise, highlighting their importance in both scientific inquiry and everyday life.